ISO 13485 certification in Saudi Arabia is beneficial to medical equipment manufacturers and providers in ensuring their Quality Management Systems (QMS) are compliant with international standards and with the SFDA regulations. It ensures the safety, performance, and traceability of products throughout the device life cycle, including the design and production, distribution, and servicing. Certification is gained from documentation reviewing and on-site audits from accredited bodies. It enhances market access (including CE marking), fosters trust from stakeholders, and supports development in Saudi Arabia’s growing healthcare system.
What is ISO 13485 Certification in Saudi Arabia?
ISO 13485 certification in Saudi Arabia helps organizations involved in the medical device lifecycle implement a compliant Quality Management System (QMS) focused on product safety, regulatory compliance, and consistent quality. It is widely used by manufacturers, suppliers, distributors, and service providers supporting medical devices in the Saudi healthcare market.
ISO 13485 Certification in Saudi Arabia – What Does It Mean and Where Is It Most Commonly Used?
ISO 13485 is a globally approved standard used in medical quality management. In Saudi Arabia, it is commonly used by these companies that are engaged in:
- Medical device manufacturing
- Importers and distributors
- Sterile and non-sterile device suppliers
- Calibration, servicing, and maintenance providers
- Companies packaging, labeling, and providing logistic support services
Saudi institutions seek ISO 13485 to increase regulatory readiness, enhance product safety, and instil confidence in health care providers and global partners.
What are the steps to get ISO 13485 Certification in Saudi Arabia?
What is ISO 13485 and Why Does it Matter for Businesses in Saudi Arabia?
ISO 13485 is designed to ensure your organization has the proper tools and processes to satisfy all stakeholder and regulatory requirements over the lifetime of a medical device. The standard includes:
- Quality management from a risk perspective
- Controlled documentation
- Complete traceability and safety of the product
- Readiness for compliance to required regulations
For Saudi Arabia companies, ISO 13485 promotes alignment with the requirements of the healthcare market and builds confidence from regulators, hospitals and international cooperation partners.
- ISO Certification Saudi Arabia
- ISO 9001 Certification
- ISO 14001 Certification
- ISO 45001 Certification
- ISO 27001 Certification
- ISO 22000 Certification
- ISO 13485 Certification
- ISO 17025 Certification
- ISO 20000-1 Certification
- ISO 22301 Certification
- ISO 31000 Certification
- CE Mark Certification
- Halal Certification
- GDPR Certification
- HIPPA Certification
We Are in Major City's
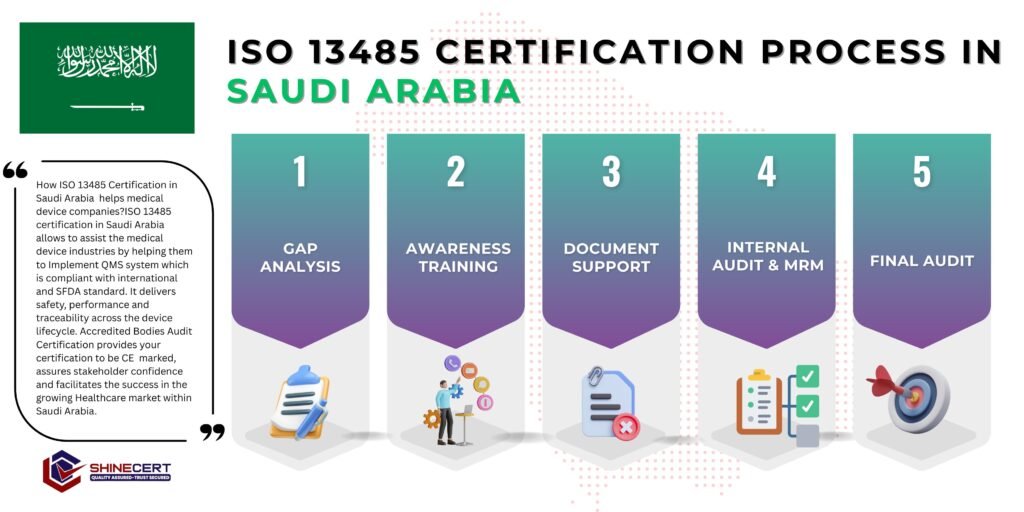
Step-by-Step ISO 13485 Certification Process in Saudi Arabia
We review your existing processes against ISO 13485 requirements and define the certification scope based on device type and activities. Common gaps include incomplete risk management files, weak supplier controls, and missing validation records.
We develop customized medical QMS documentation, including:
- Quality manual and policies
- Risk management and design controls (where applicable)
- Supplier and traceability procedures
- Complaint handling and corrective action processes
Documentation reflects real device and service activities, not generic templates.
The documented QMS is implemented across operations. We support:
- Process implementation
- Staff training on medical quality and compliance
- Alignment of suppliers and outsourced activities
This step ensures the system works in real operational conditions.
An internal audit verifies compliance with ISO 13485 requirements. Nonconformities are addressed, and management reviews system effectiveness, risks, and product performance.
An accredited certification body conducts:
- Stage 1 Audit (documentation and readiness)
- Stage 2 Audit (implementation and effectiveness)
After successful closure of audit findings, the ISO 13485 certificate is issued and remains valid for three years, with annual surveillance audits.
Benefits of ISO 13485 Certification in Saudi Arabia
Aspects of the sustained efforts that firms put into aligning themselves with the key principles of ISO 13485 include:
- Enhancement of quality and safety of medical devices.
- Improvement of process controls and traceability.
- Reduction of compliance and product risks.
- Enhanced acceptance of service delivery in the healthcare sector.
- Improved preparedness for regulatory audits.
- Gained global recognition in the medical sector.
Related ISO Certifications in Saudi Arabia
Many organizations integrate ISO 9001 with:
- ISO 9001 (quality management systems)
- ISO 14001 (Environmental Management)
- ISO 45001 (Occupational Health & Safety)
- ISO 27001 (Information Security)
- ISO 22301 (Business Continuity Management)
- ISO 50001 (Energy Management System)
- ISO 37001 (Anti-Bribery Management System)
Integrated systems reduce duplication and improve overall management effectiveness.
ISO 13485 Certification Requirements in Saudi Arabia
To achieve ISO 13485 certification, organizations must demonstrate:
- Defined quality policy and objectives
- Risk-based quality management processes
- Supplier and traceability controls
- Complaint handling and corrective action systems
- Internal audit and management review
Strong management oversight is essential for sustained compliance.
ISO 13485 Certification Cost in Saudi Arabia
The cost of ISO 13485 certification in Saudi Arabia depends on:
- Type and complexity of medical devices
- Organization size and scope of activities
- Risk classification and process controls
- Certification body audit duration
Costs include consulting support and certification body fees.
Shinecert provides transparent pricing and offers a free initial assessment.
Why Choose Shinecert for ISO 13485 Certification in Saudi Arabia?
Shinecert ISO Consulting and Certifications provides specialized ISO 13485 consulting tailored to medical device businesses.
We offer:
- Medical QMS and risk-based expertise
- Experience across device manufacturers and distributors
- Practical, audit-ready implementation
- End-to-end certification support
- Ongoing guidance for surveillance audits
Our focus is on real medical quality compliance, not documentation-only certification.
Let’s Collaborate with Us!
FAQs
Some frequently asked questions about the service that you may have questions about
ISO 13485 itself is not a license, but it is widely expected for medical device organizations to demonstrate quality system compliance.
ISO 13485 certificates are issued by accredited certification bodies. Consultants like Shinecert assist with implementation and audit readiness.
Yes. ISO 13485 is globally recognized and accepted across medical device markets.
Yes. ISO 13485 applies to manufacturers, distributors, importers, and service providers involved in the medical device lifecycle.
The certificate is valid for three years, subject to annual surveillance audits.
No. Certification depends on successful audit outcomes by the certification body. Shinecert ensures full system readiness and compliance.
Get ISO 13485 Certified in Saudi Arabia with Shinecert
If you are planning to implement a reliable quality management system, Shinecert ISO Consulting and Certifications can guide you at every step, from gap analysis to certification audit readiness.
Contact Shinecert today for a free ISO 13485 consultation in Saudi Arabia.
Request Free Consultation

