ShineCert is your trusted partner in ISO certification and consulting, o ering end-to-end services to help businesses easily achieve and maintain international standards. Specializing in ISO 13485 Certification, the leading standard for medical device quality management systems, we provide expert consultation, documentation, training, internal audits, and certification services. Our team of experienced consultants works closely with you to ensure compliance with the stringent requirements of ISO 13485 Certification Consultants, enhancing your business’s ability to deliver safe and effective medical devices. At ShineCert, we understand the unique challenges of the medical device industry and are committed to guiding you through every step of the certification process to ensure continuous improvement and regulatory compliance.
ISO 13485 Certification
ISO 13485 is the international standard for quality management systems (QMS) in the medical device industry. This standard provides a framework for companies involved in the design, production, installation, and servicing of medical devices to ensure their products meet regulatory requirements and
deliver quality and safety to users.
By implementing ISO 13485 Certification Consultants, medical device companies can enhance their ability to manage quality, reduce risk, and demonstrate compliance with global regulatory requirements. The certification is critical for companies seeking to enter or expand in markets that demand high-quality medical products, such as the European Union, the United States, and Japan.
ShineCert’s team of experts helps your organization implement ISO 13485 Certification in a way that ensures compliance, minimizes risks, and improves operational efficiencies. Whether you’re a startup or an established player in the medical device industry, we tailor our solutions to meet your specific needs, helping you gain the certification that sets your business apart in a competitive marketplace.
What are the steps to get ISO 13485 Certification?
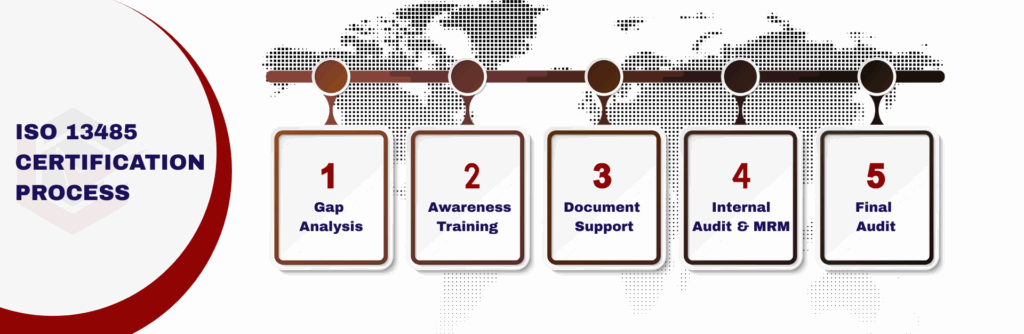
Process to Get ISO 13485 Certification
Consultation and Gap Analysis
Planning, Documentation, and Policy Development
Training and Awareness
Internal Audit and Management Review
External Certification Audit and Certification
Why ISO 13485 Certification Matters
ISO 13485 certification is essential for organizations in the medical device industry to meet regulatory requirements and improve the quality and safety of their products. Certification ensures that your medical devices meet both customer expectations and statutory requirements, reducing risks and enhancing patient safety.
Implementing ISO 13485 is a proactive approach to managing quality control, reducing product recalls, and improving the reliability of your devices. It strengthens your reputation and boosts business growth by ensuring your organization consistently delivers safe, effective, and reliable medical devices that meet or exceed regulatory standards.
ShineCert helps you streamline the process of obtaining ISO 13485 certification, ensuring that your organization meets industry standards, minimizes risks, and enhances its competitive position in the medical device sector.
Top Benefits of ISO 13485 Certification
- Regulatory Compliance
ISO 13485 helps your organization comply with international regulations such as FDA requirements (in the USA), the European Union Medical Device Regulation (MDR), and other country-specific standards, ensuring your devices meet all legal and regulatory obligations. - Enhanced Product Quality
Certification enables businesses to implement systematic quality management processes that ensure medical devices are consistently produced to meet high standards of safety and effectiveness, minimizing the likelihood of defects or non-compliance. - Improved Market Access
ISO 13485 certification is recognized globally and is often a mandatory requirement for entering international markets. Achieving certification opens the door to new business opportunities in regions like the EU, USA, and other international markets. - Risk Reduction
ISO 13485 requires companies to adopt a risk-based approach to managing product development, manufacturing, and post-market activities. This proactive risk management reduces the likelihood of product failures, recalls, and compliance issues. - Customer Confidence
ISO 13485 certification demonstrates your commitment to quality and compliance, strengthening customer trust. Customers are more likely to choose certified products from organizations that prioritize safety and quality. - Operational Efficiency: By implementing a standardized quality management system, ISO 13485 helps streamline operations, eliminate inefficiencies, and improve internal processes. This leads to cost savings, enhanced productivity, and a focus on continuous
Cost of ISO 13485 Certification
The cost of ISO 13485 certification depends on the size and complexity of your organization, the scope of your quality management system, and the number of locations involved. ShineCert offers customized pricing based on your specific needs and provides a transparent breakdown of all associated costs.
Investing in ISO 13485 certification not only ensures compliance with international regulatory requirements but also helps improve product quality and patient safety. The benefits of certification far outweigh the cost, as it enables organizations to expand their market reach, improve operational efficiencies, and reduce the risks associated with poor quality.
Why Choose ShineCert for ISO 13485 Certification
With years of experience and a strong presence in over 30 countries, ShineCert is a globally trusted partner for ISO 13485 certification. We have successfully assisted numerous organizations in achieving certification, ensuring regulatory compliance and operational excellence.
- ShineCert understands the complexities of the medical device industry and tailors its approach to meet the specific needs of each client. Our consultants have deep knowledge of the regulatory landscape and quality management practices in the healthcare sector.
Our services cover every aspect of ISO readiness, including gap assessments, documentation review, internal audits, and pre-certification checks—so you can confidently move forward.
From initial gap analysis to post-certification audits, ShineCert provides full-service support throughout the ISO 13485 certification journey. We ensure that your organization is fully prepared and confident in meeting all certification requirements.
We believe in providing honest, transparent pricing with no hidden costs. Our pricing is designed to deliver maximum value while helping you achieve ISO 13485 certification without exceeding your budget.
Ready to Get Certified?
Build trust. Ensure safety. Drive growth.
ISO 13485 certification enhances your organization’s ability to meet international standards for medical device quality management. ShineCert is here to help you through every step of the process, from gap analysis to final certification.
Take the next step in achieving ISO 13485 certification today. Call us now: +9663699403 Get a free quote: contact@shinecert.com.
Let’s Collaborate with Us!
Call us: +91 96636 99403
Frequently Asked Questions
Some frequently asked questions about the service that you may have questions about


